 Up to two electrons can occupy any one atomic orbital: one electron with a spin of s = +½ and one electron with a spin of s = -½. This is known as a “p-orbital.” This particular orbital has a quantum state of n = 2, l = 1, and m = 0 and is shaped a bit like a dumbbell, with one region in front of the nucleus and a second region behind the nucleus. The thirteenth electron of an aluminum atom actually occupies an atomic orbital that looks something like this. Neither of those elements has been named by their discoverers yet. The elements 113-118 have all been synthesized and detected in the lab, but so far, only the discoveries of ununquadium (atomic number 114) and ununhexium (atomic number 116) have been officially confirmed in June 2011. Copernicium (atomic number 112) was finally named in February 2010. (Elements that occur “naturally” are created by nucleosynthesis in a star, during supernovas, or when natural but unstable isotopes decay radioactively.)īy 1996, the first 112 elements had been identified. Originally, it was believed that uranium was the heaviest known atom that occurred naturally, but it was later discovered that neptunium and plutonium (both originally synthesized and detected in the lab) also occurred naturally but in very small quantities. These heavier elements were created by bombarding atomic nuclei with protons and neutrons in nuclear reactors or particle accelerators (machines that use electromagnetic fields to accelerate particles close to the speed of light). Nuclear research in the 1940s (including the Manhattan Project) that led to the first atomic bombs produced the first man-made elements. By 1940, all of the elements from atomic number 1-92, except promethium (atomic number 61) had been discovered and identified. There were many gaps in the table where individual elements had not yet been discovered, including fluorine (atomic number 9), neon (atomic number 10), argon (atomic number 18), krypton (atomic number 36), xenon (atomic number 54), radon (atomic number 86), and radium (atomic number 88). The lightest element was hydrogen (atomic number 1) and the heaviest element was uranium (atomic number 92). The first modern periodic table was created in 1869. Copper and chlorine are slightly unusual in that they both have two relatively common isotopes. All of the other elements listed in the table above have isotopes that are even more common (practically 100%). 98.94% of the carbon atoms on Earth are 12C, 99.64% of the nitrogen atoms are 14N, 99.76% of the oxygen atoms are 16O, 75.76% of the chlorine atoms are 35Cl, 96.94% of the calcium atoms are 40Ca, and 91.75% of the iron atoms are 56Fe. 
99.99% of the hydrogen atoms on Earth are 1H, so when we talk about the “hydrogen” atom, we usually mean 1H. It is easy to get lazy and ignore the fact that most types of atoms can exist as several different isotopes because one of those isotopes is usually far more common than the others. However, the actual mass of a mole of copper will depend on the composition of the specific sample. One mole of copper will therefore have a combination of both copper-63 atoms and copper-65 atoms in it, and the “molar” mass of copper is considered to be 63.546 g/mol. Copper-65 has a greater mass than copper-63 because it has two more neutrons in its nucleus. It has a mass number of 65 (29 protons and 36 neutrons) and an atomic mass of 64.9278 u. Most of the remaining 31% of copper atoms on Earth are copper-65 ( 65Cu). It has a mass number of 63 (29 protons and 34 neutrons) and an atomic mass of 62.9296 u. About 69% of the copper atoms on Earth are copper-63 (this percentage may vary in other solar systems and galaxies). Copper-63 ( 63Cu) is the most common isotope. Copper can exist as 29 different isotopes, and two of those isotopes are stable. There is not just one type of copper atom. Both protons and neutrons have masses approximately equal to 1 u (1 proton ≈ 1.0073 u, 1 neutron ≈ 1.0087 u, and 1 u ≈ 1.66 × 10 -27 kg), so the mass of a copper atom will be approximately 63 u, and the mass of 1 mole (6.022 × 10 23 atoms) of copper will have a mass of approximately 63 g. Because almost all of an atom’s mass is in its nucleus, we can estimate an atom’s mass just by counting its protons and neutrons. 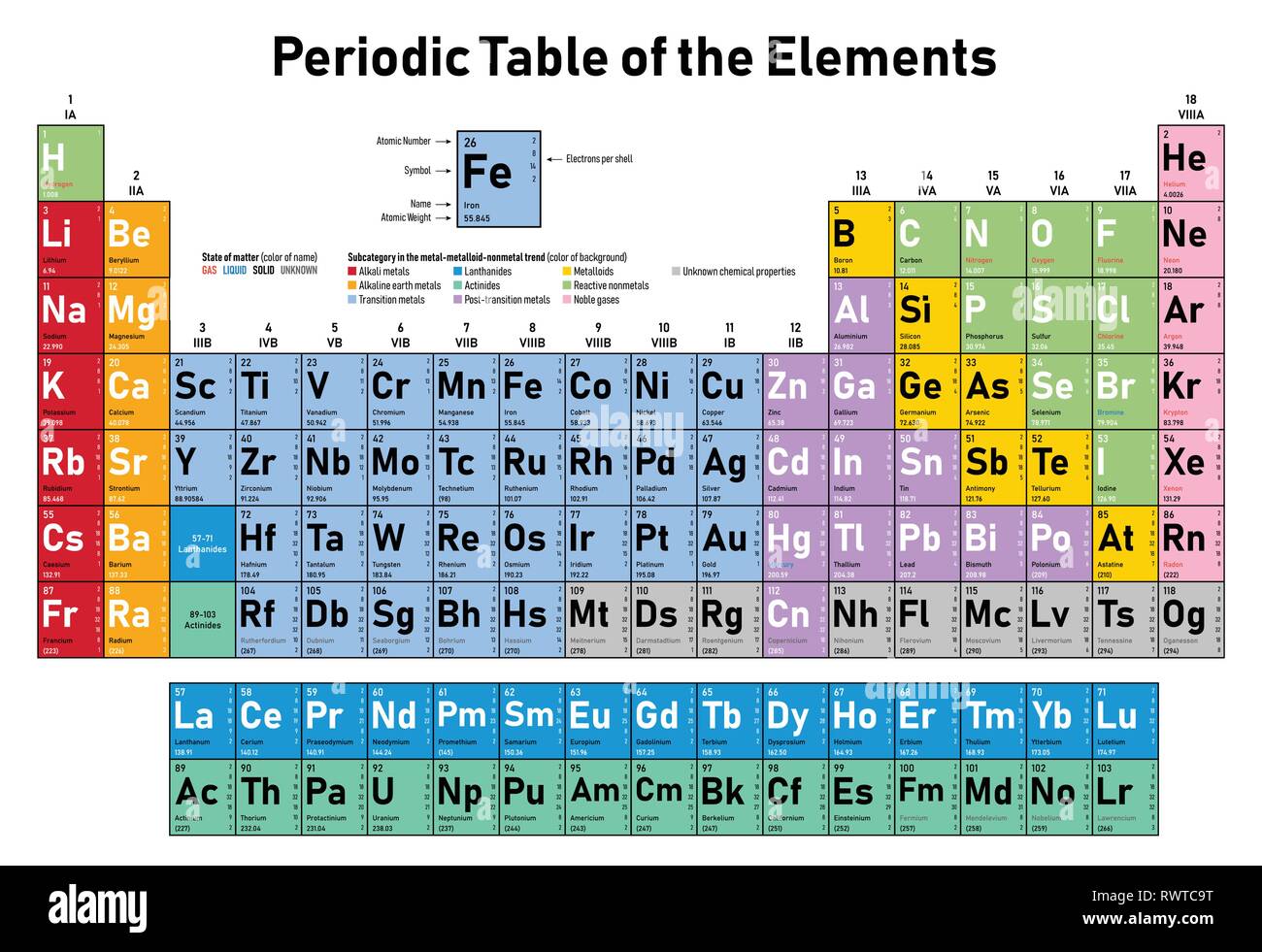
A neutrally charged copper atom will then have 29 electrons in orbit around its nucleus (29 − 29 = 0). This means that the copper atom has 29 protons and 34 neutrons in its nucleus (29 + 34 = 63). The atomic number is the number of protons in the nucleus, and the mass number is the number of protons and neutrons in the nucleus. 
Copper has an atomic number of 29 and a mass number of 63. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
